Large quantities of gases emitted to the atmosphere associated with the Holuhraun eruption
Large quantities of gases are emitted to the atmosphere associated with the Holuhraun eruption, Icelandic Met Office (IMO) reported on Thursday, November 13, 2014. Among the gases are SO2 that can convert to sulfuric acid causing acid rain.
Rainwater samples have been collected and analyzed at weekly bases at 18 locations across all the country since September 19, 2014. The sampling and analysis have been carried out by Veðurstofan and Háskóli Íslands.
The pH of unpolluted rainwater in Iceland is ~5.6. Rainwater with lower pH is referred to as acid rain and rainwater with higher pH may have interacted with dust in the air and the rain collectors.
The pH of rainwater in Iceland, after the start of the Holuhraun eruption, is ranging from pH 3.2 to 7.5 with an average value of pH 5.8. About 40% of the rain is defined as acid rain whereas 60% can be regarded as unpolluted rain. Less than 1% of the acid rain is however very acid with pH below 4. Stations at Borgir, Reykjahlíð, Írafoss, Hitardalur and Hjarðarland, are among those showing acid rain periodically, IMO said.
It is important to consider that several factors affect the pH of rainwater in addition to the volcanic eruption, the main factors being seaspray, geothermal activity, evaporation and fine rock dust.
The analysis will continue as soon as new samples will be available and the updates will be published on IMO's web-site.
Source: IMO
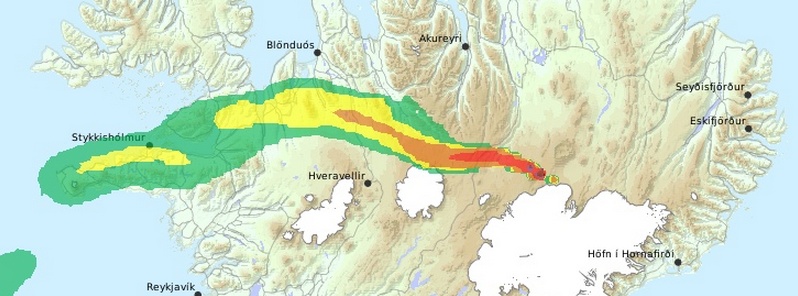
Featured image: Gas dispersion model for Friday, November 14, 2014. Image credit: IMO

Commenting rules and guidelines
We value the thoughts and opinions of our readers and welcome healthy discussions on our website. In order to maintain a respectful and positive community, we ask that all commenters follow these rules.