New type of MRSA in hospitalized patients discovered
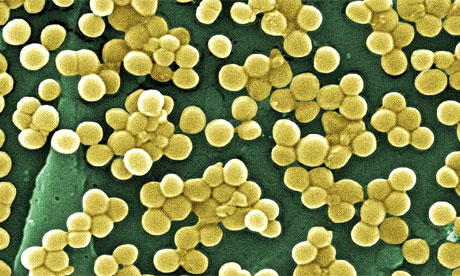
A distinctly new type of methicillin resistant Staphylococcus aureus (MRSA) that is not detected by traditional genetic screening methods has been discovered in patients in Irish hospitals according to research to be published in the journal Antimicrobial Agents and Chemotherapy. These findings provide significant insights into how new MRSA strains emerge and highlight the potential for the transmission of infectious agents from animals to humans.
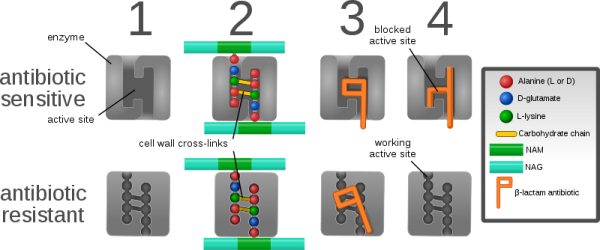
MRSA is a significant cause of hospital- and community-acquired infection worldwide. MRSA strains are characterized by the presence of a mobile DNA cassette (known as SCCmec) encoding genes that confer resistance to beta-lactam antibiotics including methicillin and recombinase genes that allow the cassette to transfer into methicillin-susceptible S. aureus (MSSA).
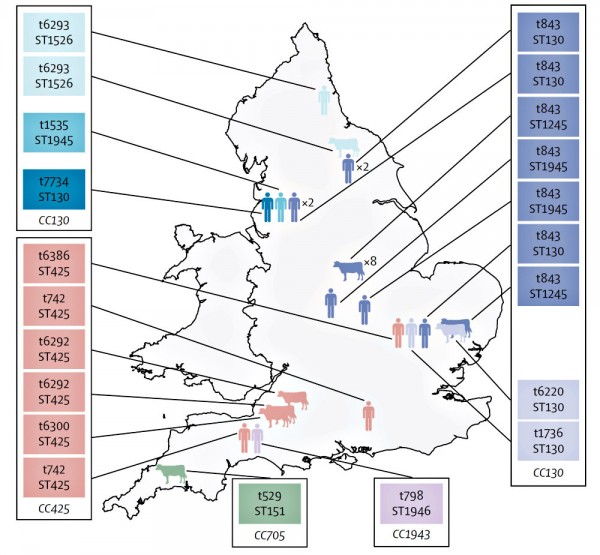
Scientists at the University of Dublin, the Irish National MRSA Reference Laboratory and the University of Dresden and Alere Technologies in Germany identified the new MRSA strain using high throughput DNA microarray screening. Complete genome sequencing revealed that this strain is distinctly different to previously described MRSA. It carries a new type of SCCmec encoding highly divergent genes that are very different to any described previously in MRSA or in any other organism. Consequently the new strain is not detected as MRSA by routine conventional and real time DNA-based polymerase chain reaction (PCR) assays commonly used to screen patients for MRSA. The MRSA strain was found to belong to the genetic lineage clonal complex 130 (CC130), which has previously only been associated with MSSA from cows and other animals, but not humans, strongly suggesting that the new MRSA originated in animals.
During the publication process, the authors became aware that a consortium of researchers lead by the University of Cambridge and the Wellcome Trust Sanger Institute in the United Kingdom had identified bovine MRSA with an almost identical SCCmec element to that in the Irish CC130 human MRSA. These researchers also identified MRSA harboring the novel SCCmec element emerging in bovine and human populations in the United Kingdom and Denmark. This study will be published simultaneously in Lancet Infectious Diseases.
Commenting on the significance of the findings, Professor David Coleman from the University of Dublin said: “The results of our study and the independent United Kingdom study indicate that new types of MRSA that can colonize and infect humans are currently emerging from animal reservoirs in Ireland and Europe and it is difficult to correctly identify them as MRSA. This knowledge will enable us to rapidly adapt existing genetic MRSA detection tests, but has also provided invaluable insights into the evolution and origins of MRSA.“ (MedicalExpress)
Provided by American Society for Microbiology

Gross! So scary, I worked at a nursing home while I was pregnant (as a housekeeper) needless to say I double gloved in those rooms